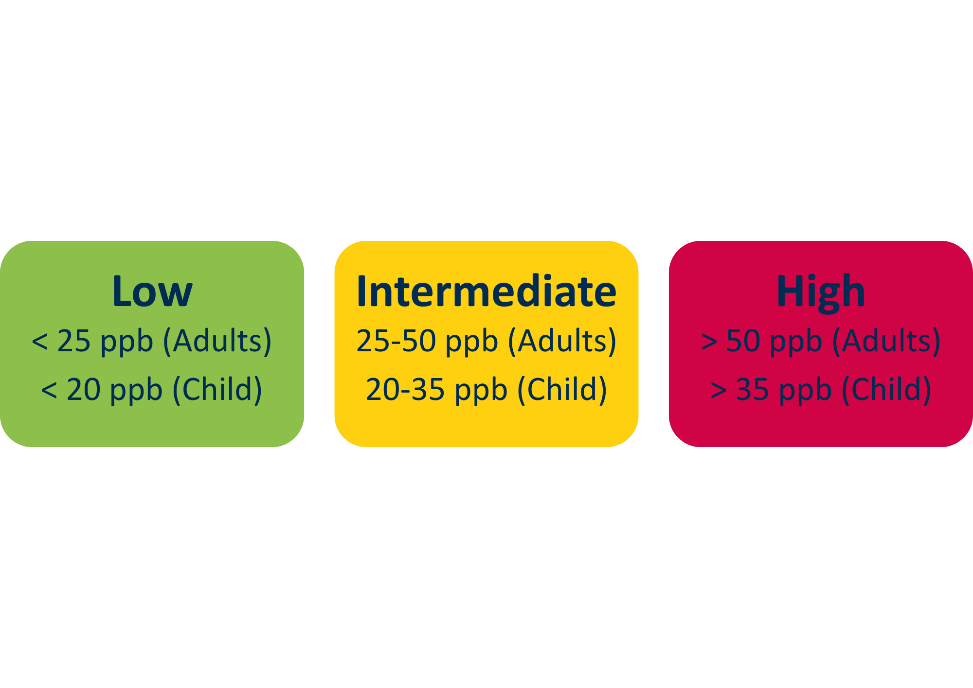
Bedfont® ensures exercise books reach UK classrooms through sponsorship.
Bedfont® Scientific Limited, a world leader in breath analysis, with nearly 50 years of expertise in the medical device industry, has teamed up with Rymindr, a social-tech company, to deliver vital classroom supplies. Rymindr is an app that lets users track important dates and access rewards. Through the app, Rymindr helps UK schools save money by sharing its profits. Bedfont® is proud to support its exercise book sponsorship campaign by sponsoring 20 boxes of exercise books, ensuring they reach the classrooms where they are needed.
It has been reported that over 70% of schools in the UK are underfunded1, leading to increased pressure on schools through teacher shortages and reduced resources. Rymindr’s mission is to build a single ecosystem that benefits all, leading to positive impacts on society and the environment for this generation and those to come. They have been funding breakfasts, school meals, and classroom supplies for children.
“We’re thrilled to partner with Bedfont® to get essential classroom supplies into UK schools. With budgets under pressure, simple items like classroom resources from exercise books to pencils shouldn’t be a struggle to fund, and this sponsorship will make a real difference in classrooms across the country.” Said Rav Panesar, CEO of Rymindr. “We’re incredibly grateful to Bedfont® for stepping up and sponsoring. This is exactly the kind of community impact we want to create through Rymindr, bringing businesses and schools together to deliver practical support where it’s needed most.”
Bedfont® is supporting their current initiative to get thousands of brand-new exercise books directly into UK primary schools. Having already distributed 4,000 boxes across the country, Rymindr were unfortunately notified at the end of 2025 that their storage facility was being sold. This has led to the launch of their latest sponsorship campaign, inviting individuals and businesses to sponsor boxes of books. The sponsorship covers the practical logistics, such as storage, pallet handling, and distribution.

“We’re incredibly proud to support this campaign and help deliver something as fundamental as exercise books to classrooms across the UK.” Said Jason Smith, CEO at Bedfont®. “With so many schools facing funding pressures, it’s vital we step in where we can. By sponsoring these boxes, we’re helping teachers focus on what matters most, teaching, while easing some of the financial strain they face every day. Education is the foundation of our future, and we’re committed to playing our part.”
Rymindr is matching each sponsored box, meaning schools across the UK are saving around £100 per box, which will really make a difference to the already stretched school budgets. Bedfont® is strongly committed to supporting the local community and is pleased to assist Rymindr in its latest initiative, providing much-needed exercise books to UK schools. To find out more about Rymindr, visit the website here. For more information on Bedfont® and its community initiatives, visit the website here.
References
- New data finds that 7 in 10 schools in England faced budget cuts since 2010 | National Education Union [Internet]. National Education Union. 2025. Available from: https://neu.org.uk/latest/press-releases/new-data-finds-7-10-schools-england-faced-budget-cuts-2010