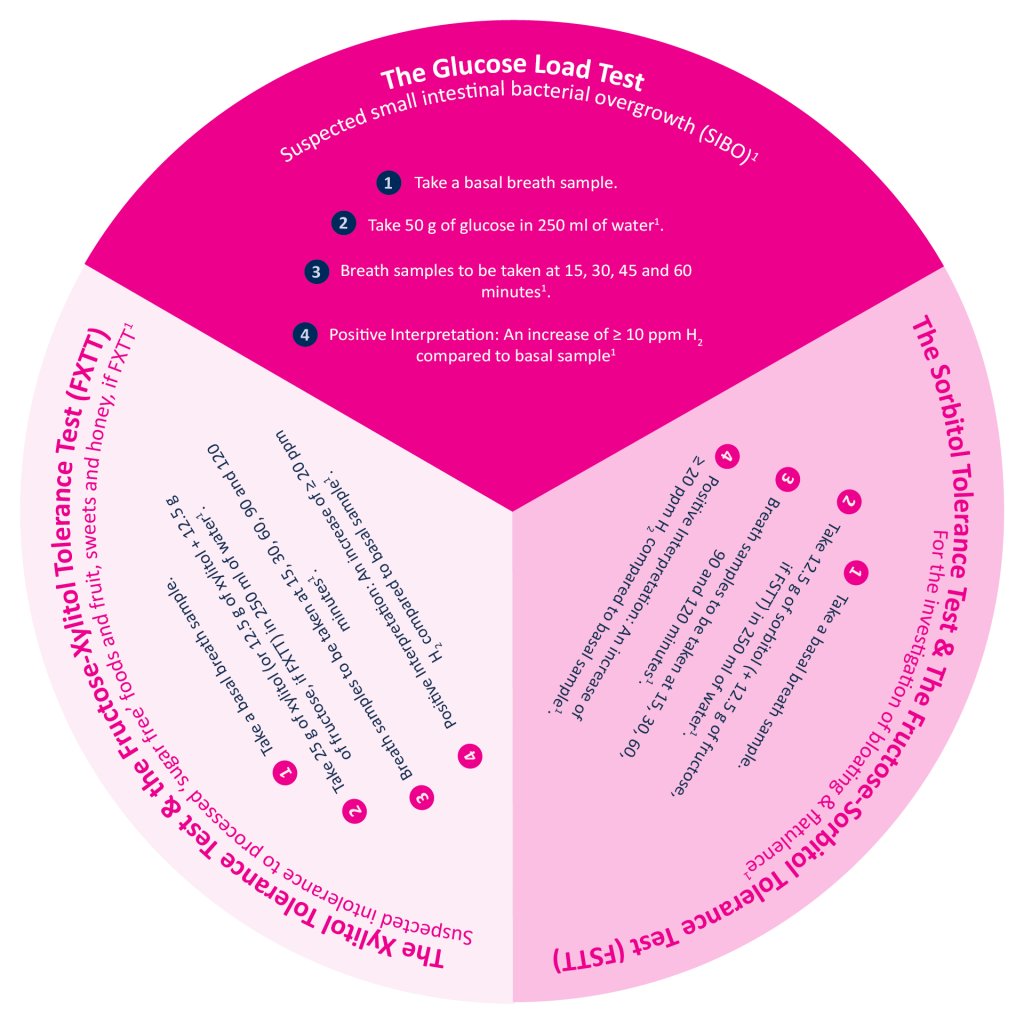
Almost 10 months after the National Institute for Health and Care Excellence (NICE), the British Thoracic Society (BTS), and the Scottish Intercollegiate Guidelines Network (SIGN) released an updated joint guideline for asthma care and management, a major milestone has been hit. Over 1 million people are now using combination inhalers instead of relying solely on blue reliever inhalers, marking a major shift towards inflammation-focused asthma management.1
Why does this matter?
Previously, many people living with asthma relied on their blue reliever inhaler, which relieved symptoms, making them feel better quickly. The problem with this was that whilst symptoms were being addressed, the underlying airway inflammation, the main driver of their symptoms, was not being treated. If the inflammation is not treated, symptoms continue to recur, damaging quality of life and increasing over-reliance on medication.1
The problem with symptom-led asthma care.
Patients who rely heavily on Short-Acting Beta-Agonist (SABA) inhalers (rescue inhalers) may report feeling well and believe that their asthma is well controlled because regular use of their inhaler keeps their symptoms at bay.1 When, in reality, this reliance is masking the underlying issue: airway inflammation.

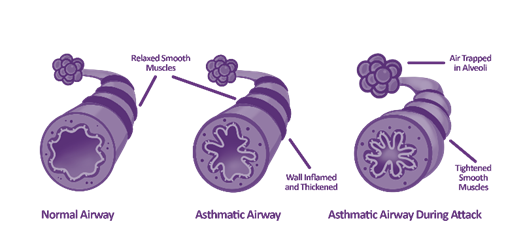
Airway inflammation cannot always be felt, and if left untreated, it could lead to further exacerbations, including asthma attacks.
The shift towards prevention-focused care.2
The update to the guideline placed greater emphasis on treating the underlying inflammation rather than just the symptoms, demonstrating an evolution in asthma care. The updated guidance reflects:
- Earlier anti-inflammatory treatment,
- Reduced SABA reliance,
- Prevention of exacerbations,
- More proactive asthma management.
The impact of this guidance is clear: over 1 million people now use combination inhalers to relieve symptoms and treat underlying inflammation, a significant milestone for asthma care.1
Why objective insight matters.
During routine asthma reviews, patients are typically asked about their symptoms, which rely on patient recall. Typically, people with asthma downplay their symptoms, believing that what they experience is typical of their condition.3
And whilst peak flow remains a useful tool in asthma care, it alone may not provide the full picture. It measures airflow obstruction, rather than the inflammatory activity driving asthma, demonstrating the need for objective assessment, because better asthma care starts with better insight.
The role of FeNO testing.2
Fractional exhaled Nitric Oxide (FeNO) testing provides an objective tool to measure airway inflammation. It is non-invasive, easy to do at the point of care and supports:
- Diagnosis,
- Inhaled corticosteroid (ICS) decisions,
- Adherence assessment,
- Monitoring,
- Evaluation of exacerbation risk.
Aligning with the latest guidance, FeNO supports the move towards more personalised, biomarker-driven asthma care.
The NObreath® FeNO device.
The NObreath® is a modern FeNO device; it is portable and easy to use, making it perfect for primary care. Clinicians can measure a person’s airway inflammation in a short amount of time, fitting perfectly into a standard healthcare appointment. This objective insight enables clinicians to make informed decisions there and then without the need for a follow-up appointment to discuss results.
Combined with an extensive clinical history, a FeNO test with the NObreath® can aid in an asthma diagnosis and tailor treatment appropriately, providing enhanced asthma monitoring.
Precision medicine and the future of asthma care.2
Guidelines are updated as technology and research evolve to improve patient care. The latest guideline from NICE/BTS/SIGN marked a move towards:

- Precision medicine,
- Biomarkers
- Personalised pathways
- Earlier intervention,
- Better outcomes.
The move towards combination inhalers marks more than a treatment change; it represents a broader evolution in how asthma is understood and managed. Objective insight can provide clinicians with a better understanding of what is going on beneath the surface, with FeNO testing supporting this objective insight and supporting future asthma care.
As asthma care continues to evolve, understanding airway inflammation will become increasingly important in helping clinicians deliver a more personalised and proactive approach.
To learn more about the NObreath® device and how FeNO testing is improving patient outcomes worldwide, visit the website here.
References
- “I no longer feel asthmatic”: more than a million people now using new style inhalers [Internet]. NICE website: The National Institute for Health and Care Excellence. NICE; 2026. Available from: https://www.nice.org.uk/news/articles/-i-no-longer-feel-asthmatic-more-than-a-million-people-now-using-new-style-inhalers
- National Institute for Health and Care Excellence. Overview | Asthma: diagnosis, monitoring and chronic asthma management (BTS, NICE, SIGN) | Guidance | NICE [Internet]. Nice.org.uk. NICE; 2024. Available from: https://www.nice.org.uk/guidance/NG245
- Bidad N, Barnes N, Griffiths C, Horne R. Understanding patients’ perceptions of asthma control: a qualitative study. European Respiratory Journal. 2018 May 17;51(6):1701346.