In 2019, 262 million people worldwide had asthma, with 455,000 deaths from the chronic condition1, stark figures that highlight asthma control still needs improvement. Most people living with asthma believe that no physical symptoms mean their asthma is controlled, but this is sadly not always the case. Airway inflammation is the underlying cause of asthma, something that cannot be seen and not always felt.
Many asthma patients feel fine until they don’t. Objectively measuring airway inflammation with Fractional exhaled Nitric Oxide (FeNO) testing is a vital way to prevent exacerbations and improve overall asthma care.
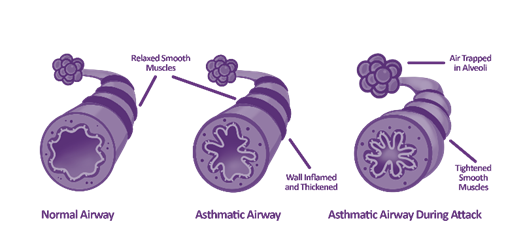
What is FeNO and why does it matter2?
When airways are inflamed, they produce excess nitric oxide (NO), a gas that can be measured by performing a FeNO test. A FeNO test provides insight into a person’s airway inflammation by measuring NO levels in exhaled breath.
- High FeNO levels indicate eosinophilic airway inflammation. The higher the reading, the more severe the inflammation is.
By measuring airway inflammation, healthcare professionals can use the results to aid an asthma diagnosis and tailor treatment appropriately.
Limitations of traditional asthma monitoring.

Traditionally, clinicians have relied on patients relaying their symptoms, which doesn’t always give the full picture of a person’s asthma. Many people believe that because they have asthma, they should have a cough and they should have a wheeze, and they downplay their symptoms.
Providing a FeNO test at regular asthma reviews empowers healthcare professionals to detect airway inflammation early, allowing them to monitor medication response and personalise treatment. This ultimately can catch an exacerbation early and get patients back on track with well-controlled asthma.
Additionally, medication adherence rates range from 30% to 70%3. A patient may report taking their medication regularly and using the correct technique; however, a quick, easy FeNO test can reveal poor adherence. Showing a patient their airway inflammation can create an opportunity to discuss and reinforce the importance of good adherence and technique.
Recent advancements in FeNO testing.

In the 1990s, researchers found that FeNO served as a non-invasive biomarker of airway inflammation and used chemiluminescence to measure it. This method proved very costly due to regular maintenance and calibration, and difficult to access because of the machines‘ size and the specialist training required.
In recent years, this has advanced to electrochemical sensor technology, enabling more cost-effective, portable FeNO devices like the NObreath®, whilst still matching the gold-standard technology of chemiluminescence. Thanks to continuous innovation, FeNO devices are now accessible at the point of care and provide instant results, improving patient outcomes.
To learn more about chemiluminescence and electrochemical sensor technology, read our article here.
In the late 1990s, FeNO began to appear in international clinical practice guidelines for asthma, and the American Thoracic Society (ATS) officially recommended its use to assess airway inflammation in adults and children in 20114.
Since then, FeNO testing has been recommended in asthma guidelines across the world, with the most recent joint guidance from the National Institute for Health and Care Excellence (NICE), the British Thoracic Society (BTS), and the Scottish Intercollegiate Guidelines Network (SIGN), recommending FeNO as a first-line test for asthma diagnosis5.
To learn more about the global guidelines on asthma care and management, read our article here.
The NObreath® FeNO device
The NObreath® was developed in 2008, designed with healthcare professionals and patients in mind. It provides a non-invasive, fairly priced option for FeNO testing, making it more accessible to healthcare providers worldwide.
Featuring an adult and child test mode, the NObreath® is suitable for all settings thanks to its accuracy and portability. It requires an exhalation-only manoeuvre, making it a quick and easy test to take for both adults and children.
The device was designed with the patient in mind and features multiple flow incentive screens. These incentive screens not only engage the patient, but they also ensure a correct flow rate of 50 ml/s, providing an accurate test result.
To learn more about FeNO testing with the NObreath®, visit the website here.

Many deaths from asthma are thought to be preventable, meaning better asthma care is needed. Advances in FeNO testing have been instrumental in improving asthma control, and with increasing recognition in global asthma guidelines, FeNO testing can make a difference.
To learn more about Bedfont® Scientific Limited, world leaders in breath analysis with over 49 years of expertise in the medical device industry, visit the website here.
References
- World Health Organization. Asthma [Internet]. World Health Organization. 2024. Available from: https://www.who.int/news-room/fact-sheets/detail/asthma
- NHS England. NHS Accelerated Access Collaborative» Fractional Exhaled Nitric Oxide (FeNO) [Internet]. www.england.nhs.uk. 2025. Available from: https://www.england.nhs.uk/aac/what-we-do/innovation-for-healthcare-inequalities-programme/rapid-uptake-products/fractional-exhaled-nitric-oxide/
- Improving adherence in Asthma treatment | Cambridge Network [Internet]. Cambridgenetwork.co.uk. 2024. Available from: https://www.cambridgenetwork.co.uk/news/improving-adherence-asthma-treatment
- Dweik RA, Boggs PB, Erzurum SC, Irvin CG, Leigh MW, Lundberg JO, et al. An official ATS clinical practice guideline: interpretation of exhaled nitric oxide levels (FENO) for clinical applications. American journal of respiratory and critical care medicine [Internet]. 2011;184(5):602–15. Available from: https://www.ncbi.nlm.nih.gov/pubmed/21885636/
- National Institute for Health and Care Excellence. Overview | Asthma: diagnosis, monitoring and chronic asthma management (BTS, NICE, SIGN) | Guidance | NICE [Internet]. Nice.org.uk. NICE; 2024. Available from: https://www.nice.org.uk/guidance/NG245

